Molecular pathogenesis of cardiomyopathies
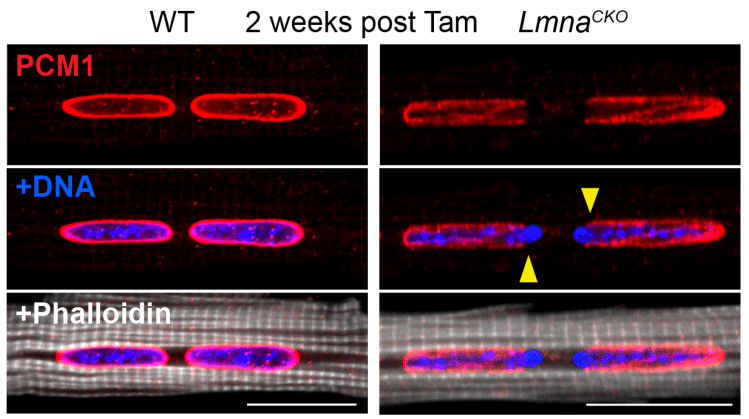
Mutations to LMNA, the gene encoding the nuclear lamina component Lamin A/C, cause severe dilated cardiomyopathies in human that often require heart transplantation. LMNA mutations are thought to be responsible for 5-10% of all familial cases of dilated cardiomyopathies. How do defects in Lamin A/C expressed almost every differentiated cell in our body cause specific disease in the heart? We investigate the molecular basis for LMNA-related cardiomyopathies using mouse models and iPSC differentiation.
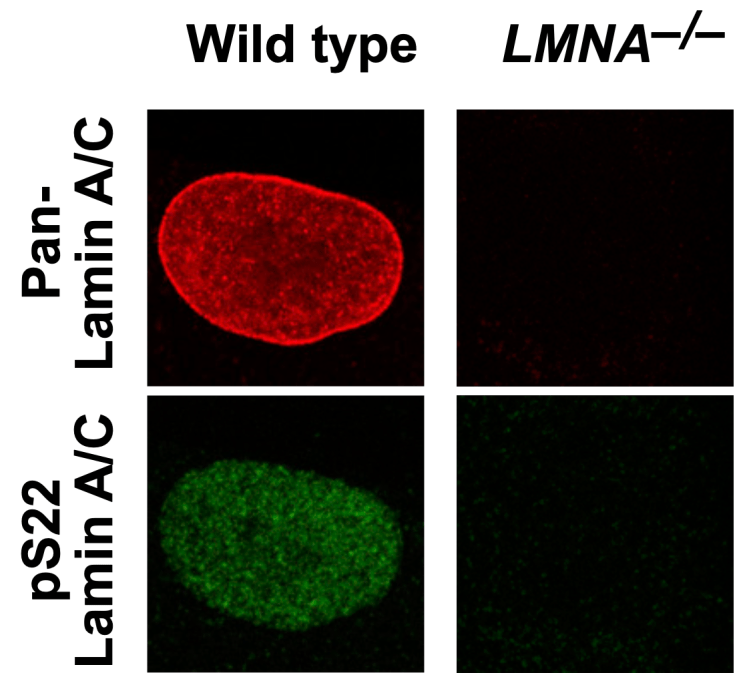
Nuclear-interior phosphorylated lamins
Nuclear lamins polymerize to form the nuclear lamina, a protein meshwork underlying the nuclear membrane. The nuclear lamina binds megabase-wide transcriptionally inactive heterochromatin domains. This project explores a new hypothesis that phosphorylated nuclear lamins localized in the nuclear interior act as transcription activators, contrary to the prevailing view that nuclear lamins are heterochromatin-binding proteins.
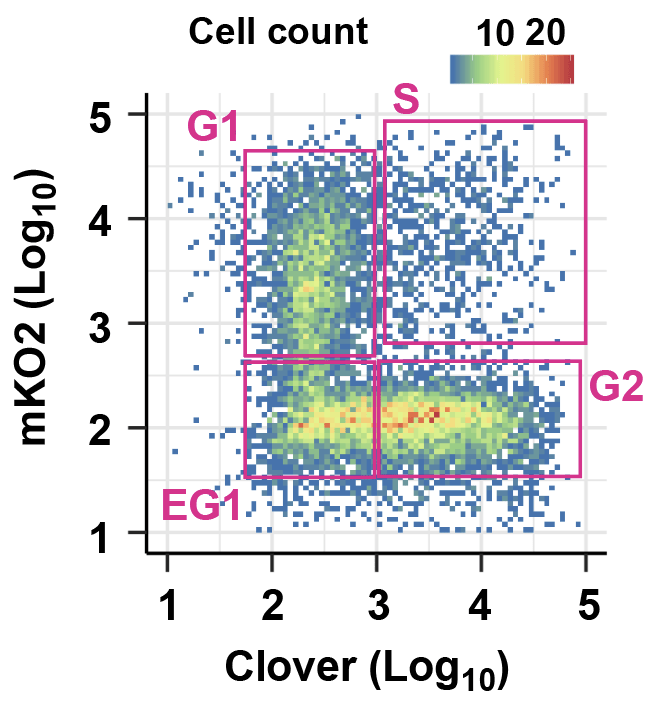
Cell-cycle dynamics of transcription
Cell-type specificity of gene expression programs is central to cell-type specific functions of the cells, and therefore, the molecular basis of multicellularity. How cell-type specific transcriptional program is maintained during the cell cycle and inherited over cell generations remains an unsolved question. This project investigates the mechanisms regulating cell-cycle dynamics of gene transcription by defining the dynamics of the chromatin state during the cell cycle and cell division.
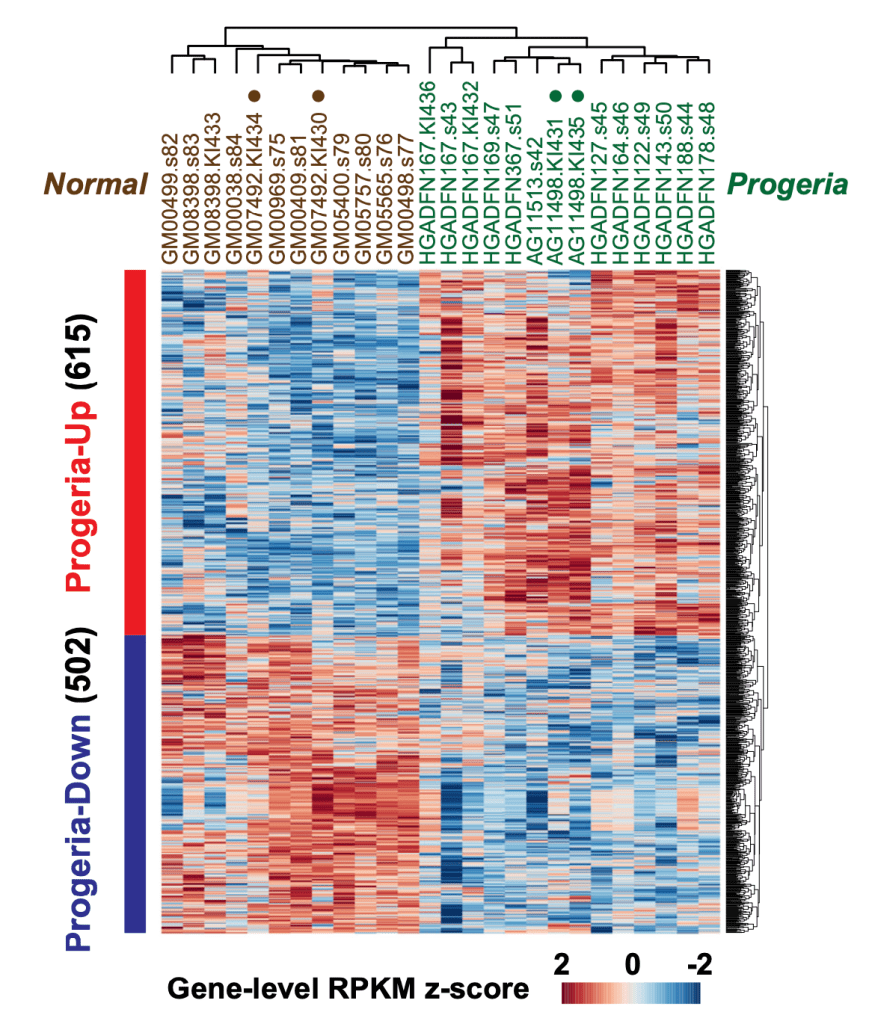
Pathogenesis of Hutchinson-Gilford progeria
Hutchinson-Gilford progeria is a rare disease often called “premature aging” disorder, caused by mutations to the nuclear Lamin A/C gene. The prevailing hypothesis for the pathogenesis of progeria is that progerin, the mutant Lamin A/C protein expressed in the patient cells, accumulates at the nuclear periphery and this causes heterochromatin disorganization, leading to derepression of genes in heterochromatin. We explore an alternative hypothesis that disrupted phosphorylated Lamin A/C-enhancer binding leads to pathogenic gene expression in euchromatin.
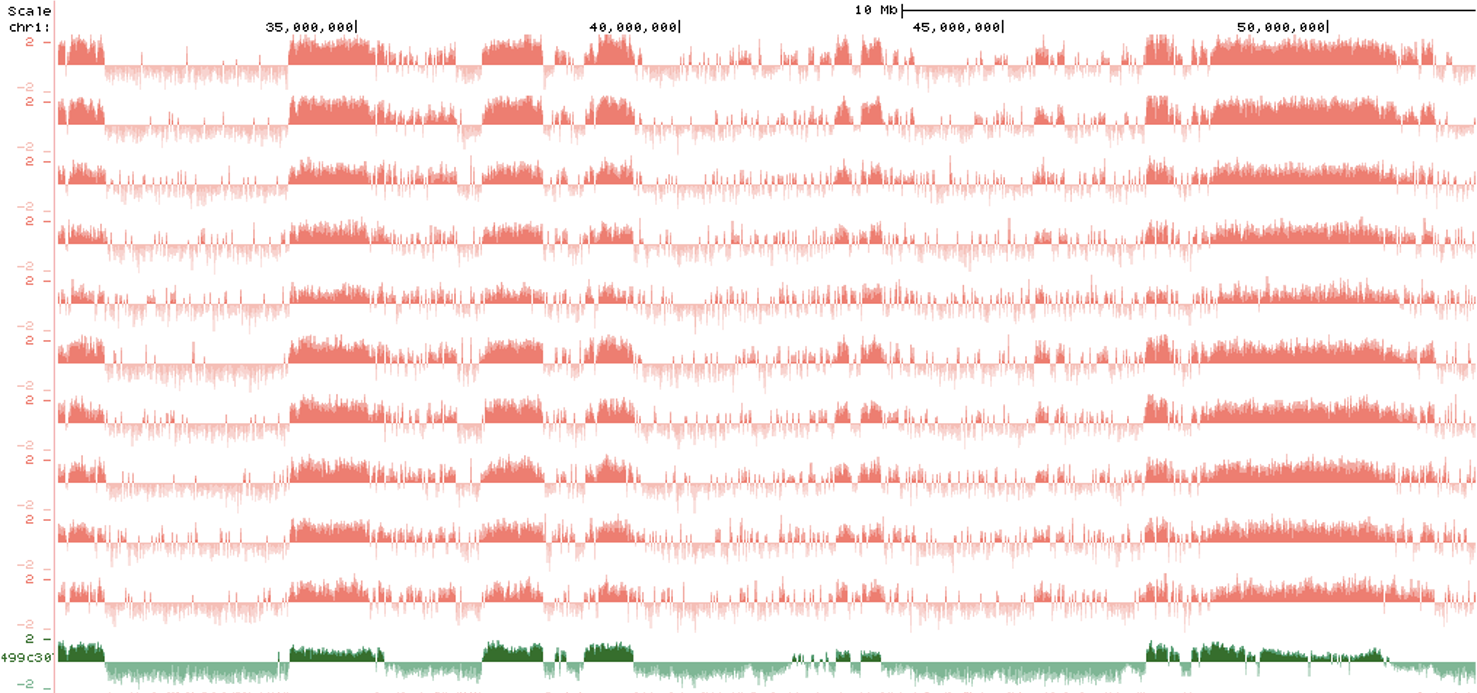
The structural organization of chromosomes by the nuclear lamina
The nuclear lamina binds heterochromatin domains called lamina-associated domains. By doing so, the nuclear lamina is thought to contribute to the spatial organization of chromosomes within the nucleus. This project uses a cell engineering approach to modulate nuclear lamin protein levels rapidly to discern the requirement of nuclear lamins for organization of chromosomes.
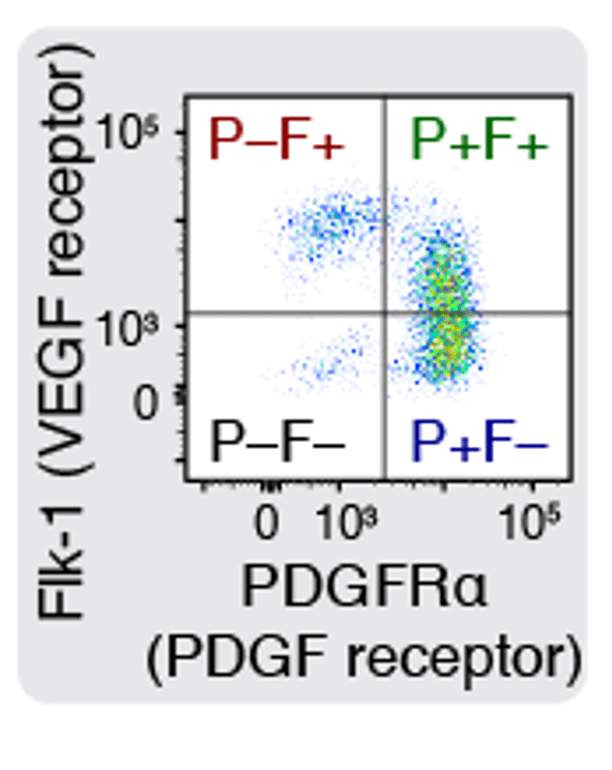
Chromatin basis for cell fate specification
Cell fate specification is the basis for the organogenesis in multicellular organisms. How the fundamentally stochastic behaviors of molecules inside and outside our cells can regulate cell fate specification in the right place at the right time? This project uses mouse embryonic stem cell differentiation to the hemato-endothelial progenitor as a model system to study how interactions between transcription factors and chromatin regulate cell fate specification process.
Chromatin landscape of cellular quiescence
Cellular quiescence is a reversible cell-cycle arrest state during which cells exit the cell cycle and enter a non-dividing phase. Maintenance of cell quiescence is fundamental to functions of terminally differentiated cells and maintaining adult stem cell reservoirs. Furthermore, modulation of quiescence allows tissue regeneration, and dysregulation of quiescence could underlie tumorigenesis. This project investigates the chromatin basis of cellular quiescence.